CNODES Query Process
Project Guide
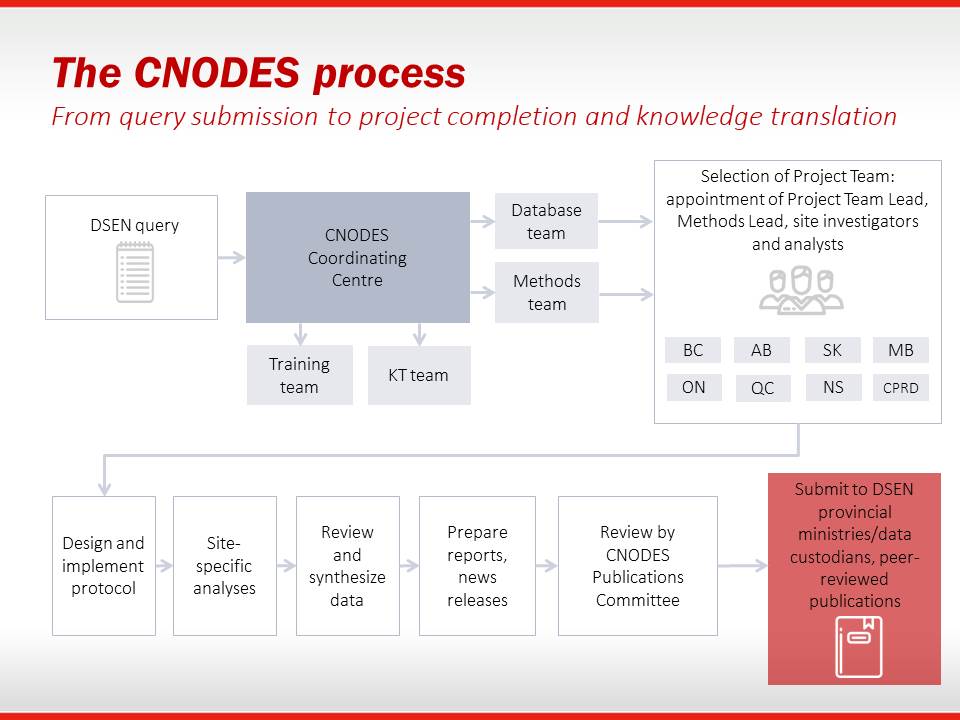
CNODES has established a standardized process to ensure the efficient progress of projects addressing important questions on drug safety and effectiveness. The Project Guide describes this process and has been put together to guide Project Team members through the steps and timelines of completing such projects. Currently under revision.
Publications Policy
This document describes the policy that CNODES has established for the authorship and acknowledgement of its contributors on all publications and presentations. It also describes the processes for the internal review of CNODES publications. Under Revisions
COI Policy
CNODES policy requires all investigators, team members, and authors of CNODES papers to fully disclose any potential conflicts of interest (COI). This document guides CNODES researchers through the identification and management of their perceived or actual COI. Under revision.